
An over-the-counter brand of eyedrops has been linked to permanent vision loss due to a bacterial infection as reported by Centres for Disease Control and Prevention.
The majority of people afflicted said they used EzriCare Artificial Tears without preservatives before getting sick.
Eleven people had eye infections, and three of them at least lost vision in one eye. Others suffered from urinary tract infections or respiratory illnesses. When the bacteria got into the patient's circulation, one individual died.
California, Colorado, Connecticut, Florida, New Jersey, New Mexico, New York, Nevada, Texas, Utah, and Washington were the states where cases were documented.
Unknown underlying eye diseases, such as glaucoma or cataracts, which would have rendered the afflicted individuals more vulnerable, may have also existed in the patients. Pain, swelling, discharge, redness, fuzzy vision, sensitivity to light, and the sensation that something foreign is lodged in the eye are all signs of an eye infection.
While the infections have not been definitively traced to the eye drops, the CDC recommended that “patients immediately discontinue the use of EzriCare Artificial Tears until the epidemiological investigation and laboratory analyses are complete.”
The eyedrops under investigation are advertised as being preservative-free. This means that nothing in the product may possibly hinder the growth of microorganisms. When a person who had the germs on their skin opened the container or during the production process, the product may have become contaminated.
The CDC discovered the bacteria in the eyedrops bottles and is conducting tests to see whether this bacteria matches the type discovered in patients.
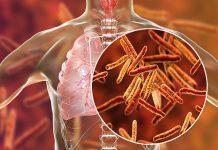
Pseudomonas aeruginosa, a kind of bacteria that is resistant to the majority of medicines, has also been found in at least 50 patients in 11 states so far, according to the CDC team.
Pseudomonas aeruginosa bacteria are commonly found in water and soil and even on the hands of otherwise healthy people. Infections usually occur in hospital settings among people with weakened immune systems.
This type of bacterium is often resistant to standard antibiotics.
“That's what's so concerning. Our standard treatments are no longer available to treat this infection,” said Dr Jill Weatherhead, an assistant professor of tropical medicine and infectious diseases at the Baylor College of Medicine in Houston.
As of Tuesday, EzriCare Artificial Tears had not been recalled. They have been sold on Amazon and at stores such as Walmart.











[…] post Eyedrops Brand Possibly Linked with Bacterial Infections, CDC Warns appeared first on […]